
The need to display Data Matrix codes on products, packaging and labels is due to a number of advantages. First of all, it is an opportunity to control the movement of a product unit from the moment of production to sale and write-off. Also, due to the use of barcode it is possible to exclude the presence of counterfeit products on the market and provide the opportunity for medical organisations to work with quality equipment and materials. In addition, labelling gives a guarantee to the buyer that he is buying a product that will not cause harm to the body.
Specialists of UCEC «QUALITY» LLC have been working for a long time in the field of preparation of documentation for registration procedures. We keep track of all changes and innovations in legislation, which gives high chances of successful completion of all stages of registration in the Honest Mark labelling system. Thanks to competent legal support from our employees, you will have an opportunity to significantly facilitate the process of mandatory labelling of certain goods intended for health purposes.
Further in the article we will consider the terms of labelling, the algorithm of actions and the types of products subject to mandatory barcodes.
Labelling of medical devices from 2023
From 2023, labelling of medical devices is among the mandatory procedures in Russia. Its introduction is carried out in several stages, which ensures the possibility of adaptation and proper preparation for all participants in the turnover.
In general, the implementation of labelled products is characterised by increased complexity. For this purpose, it is necessary to record each stage in the Honest Mark system, as well as to reflect it in product accounting.

It is more comfortable to carry out activities in one place. Inform about the withdrawal of codes from circulation; perform the exchange of documentation with organisations involved in the supply of products via electronic document management (EDI); accept information on profits and balances. All this is possible with the help of the trade automation programme MojSklad. Through it it is possible to keep records of different products. At the same time, there is a CRM and a cashier’s place in the package. Round-the-clock support, a free tariff and a quick start are provided.
Law on labelling of medical devices in Russia
From 1 September 2023, labelling of medical devices becomes a mandatory procedure, in accordance with Russian Government Decree No. 894. The document makes it possible to control the work of all participants: manufacturers, importers and retail sellers.
Mandatory labelling of medical devices with means of identification
According to the above Resolution, participants in the turnover of certain types of medical devices send an application for registration procedures to the State Information System for Monitoring the Turnover of Goods to be labelled with mandatory means of identification. These Rules became effective on 1 September 2023.
Participants in the turnover of certain types of medical devices which have a registration certificate for a product intended for health-improving use must affix means of identification to the packaging, including the possibility of affixing a label with means of identification.
In the following, we present medical devices subject to labelling on mandatory grounds:
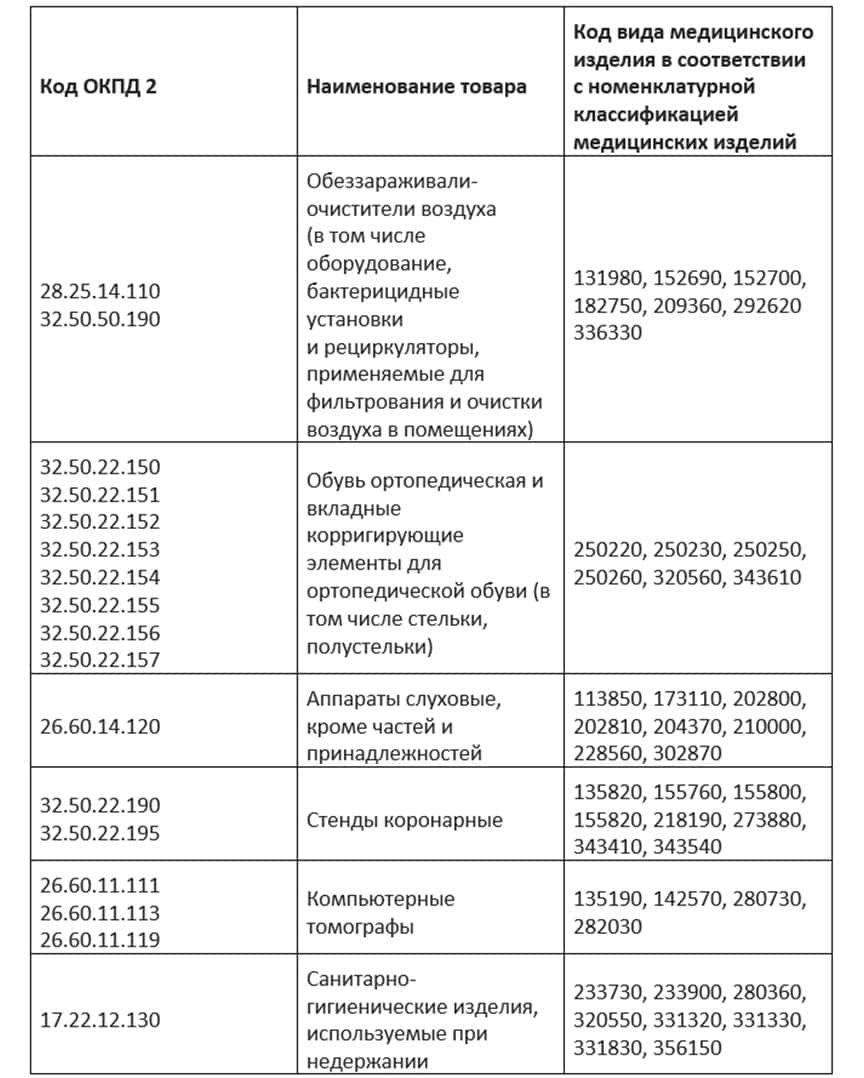
The product is labelled in case it falls under 2 years of code at once: TN VED of the EAEU and OKPD-2.
DataMatrix digital code
The DataMatrix standard stores information about each product item, as well as cryptographic security methods that eliminate the possibility of code tampering. This is the concept on which the digital product labelling system is based.
The DataMatrix code must be applied to each product item at the time of import or during the manufacturing process.
Example code:

The labelling code contains the following information:
- GTIN commodity code consisting of 14 digits;
- ISN of 13 digits;
- A code to verify the uniqueness of the labelling code.
To apply the code on the lid or label, the surface must be 13×13 mm. The exact application area and surface area are established after the experiments are performed. The price of the service related to the traceability of the labelled product is set by Russian Government Resolution No. 577 of 8 May 2019, and equates to 50 kopecks excluding VAT.
Whose responsibility is it to label medical devices?
The labelling of medical devices is performed by the manufacturer and is accompanied by the receipt of codes in the Honest Mark system. They are printed and fixed on the product with special adhesive. When interacting with a retail or wholesale buyer, a universal transfer document with the codes is created. This is how the product is put into circulation.

In addition, the labelling of medical devices must be carried out by importers before going through the customs process. That is, the goods are imported into Russia with the mandatory information (codes) applied. For this purpose, labels with Data Matrix codes are transferred to the warehouse or production workshop. A universal transfer document is also created for the next realisation.
Timing and procedure for introducing mandatory labelling of medical devices
In accordance with PRRF No. 894 dated 31.05.2023:
- On 1 September 2023, the mandatory registration procedure for all participants of the turnover in the Honest Mark system was launched;
- On 1 October 2023, compulsory labelling of medical devices began: bactericidal irradiators; footwear to correct musculoskeletal disorders of the feet with inlay corrective devices. At the same time, importers and manufacturers provide information on labelling and launch of products into circulation;
- From November 1, 2023, all importers are required to submit application report data before submitting a declaration for the following products: bactericidal irradiators; shoes that allow you to eliminate disorders in the musculoskeletal system of the legs, with inserted corrective devices;
- Until November 1, 2023, as part of the customs process carried out in relation to products imported from countries other than the EAEU member countries, it is possible that there will be no labeling codes. But after passing the Federal Customs Service, it is necessary to label each product unit, as well as provide information about the launch into circulation on a mandatory basis;
- On March 1, 2024, it is planned to label certain types of medical products: CT devices; devices intended to be installed in the coronary arteries and supply the heart; Hearing Aids; devices used for sanitary and hygienic purposes; devices used for involuntary loss of urine. At the same time, manufacturers and importers are required to provide data on the application of codes and the launch of products into circulation;
- On April 1, 2024, all importers are required to submit application report data before submitting a declaration for products: devices intended for installation in the coronary arteries and cardiac supply; Hearing Aids; devices used for sanitary and hygienic purposes; devices used for involuntary loss of urine;
- Until April 1, 2024, during the process of passing customs control carried out in relation to products imported from countries other than the EAEU member countries, it is possible that there will be no labeling codes. But after passing the Federal Customs Service, it is necessary to label each product unit, as well as provide information about the launch into circulation on a mandatory basis;
All remaining unsold products belonging to the above types of health-related goods, having an operational life, manufactured or imported into the Russian Federation before the launch of mandatory labeling, must be marked with the appropriate codes before August 31, 2024.

